
Patient Support
Latest News
Latest Videos

More News

A panel discussion breaks down ways for stakeholders to meet temperature-controlled operational demands pertaining to product pipelines.

In the second part of her video interview with Pharma Commerce Editor Nicholas Saraceno, Rachel Thorpe, executive director at the Otsuka Patient Assistance Foundation, lays out the ways to begin transitioning a foundation.

In the second part of her video interview with Pharma Commerce Editor Nicholas Saraceno, Kimberly Westrich, chief strategy officer at the National Pharmaceutical Council, describes how accumulators, maximizers, and AFPs are impacting patients, manufacturers, and healthcare ecosystems, and how that all ties into the presentation she participated in.

In the third part of his video interview with Pharma Commerce Editor Nicholas Saraceno, Colin Banas, MD, DrFirst’s chief medical officer, uncovers the main reasons why 52% of respondents choose DTC online prescription platforms.
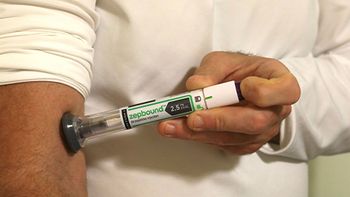
The telehealth company is offering Lilly’s branded form of tirzepatide—approved for weight loss—on its site for $1,899 a month, while Lilly also sells the GLP-1 via its LillyDirect platform.

Renier Brentjens, MD, PhD outlines the evolution of the space, while offering ways to boost patient access to these treatments.

The tool will focus on educational resources by offering both in-person and telehealth support for individuals living in the United States.

A Day 3 presentation dives into the impact of copay accumulators, maximizers, and AFPs on driving cost savings.

The presentation suggests ways to educate patients on the options available to them, with the hope of ultimately improving health outcomes.

The session provides an overview of the uninsured population, and also tackles challenges faced by patients when it comes to accessing PAPs.

A roundtable explores how patient access needs are being measured, while also discussing the value of affordability in decision-making.

The conference’s first panel offers insight on the current state of awareness among beneficiaries of the Part D cap and Medicare Prescription Payment Plan via the PAN Foundation’s latest research.

The conference’s opening session highlights the sector’s current landscape of healthcare coverage and access.

In the third part of his video interview with Pharma Commerce Editor Nicholas Saraceno, Jason C. Foster, CEO of Ori Biotech, discusses how regulatory hurdles are impacting the advancement of CGTs, along with the changes he would like to see along that front in order to accelerate patient access.

The partnership focuses on enhancing the patient experience by providing greater affordability and access to specialty meds.

In the first part of his video interview with Pharma Commerce Editor Nicholas Saraceno, Jason C. Foster, CEO of Ori Biotech, offers his perspective on the state of CGTs, along with their potential for transforming medicine.

In the final part of his video interview with Pharma Commerce Editor Nicholas Saraceno, Tom Dorsett, CEO, RazorMetrics, describes how the software company’s solution can help deliver better patient outcomes and substantial savings for healthcare plans, employers, and members alike.
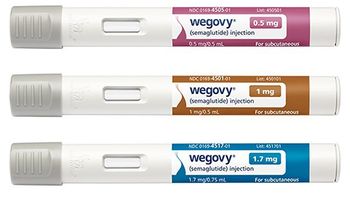
NovoCare will offer the GLP-1 injection to cash-paying patients for a discounted $499 per month.

In the final part of his video interview with Pharma Commerce Editor Nicholas Saraceno, Geoffrey Joyce, PhD, director of health policy at the Leonard D. Schaeffer Center for Health Policy & Economics at USC, describes how PBMs financially benefit from generics, and why a completely open generic market powered by cash would be beneficial.

Why meaningful content must meet the right audience.

The presidential action follows up on a 2019 final rule aimed at empowering patients and boosting competition among hospitals, group health plans, and health insurance issuers in both individual and group markets.

Patients who utilize the self-pay pharmacy will also be receiving savings on select sizes.

In the second part of his video interview with Pharma Commerce Editor Nicholas Saraceno, Tom Dorsett, CEO, RazorMetrics, comments on why he believes polypharmacy is an overlooked driver of unnecessary spending and adverse health outcomes.

In the first part of his video interview with Pharma Commerce Editor Nicholas Saraceno, Tom Dorsett, CEO, RazorMetrics, explains the concept of polypharmacy, including how prominent it is in the United States today.

James D. Chambers, PhD, MPharm, MSc, professor of medicine at the Tufts Medical Center Institute for Clinical Research and Health Policy Studies, dives into why biosimilars contribute to a reduction in cost and increase in patient access, while highlighting challenges to adoption.














