
Outsourced services can provide significant financial and risk-avoidance benefits

Outsourced services can provide significant financial and risk-avoidance benefits
Good growth is occurring in the vaccine business, despite the hurdles of public perceptions and public-health funding shortfalls


The rise in product volume is more than matched by the rise of regulatory constraints
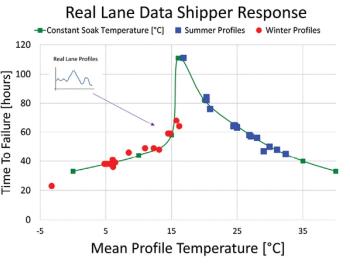
A 'performance curve' chart for containers could simplify the design process
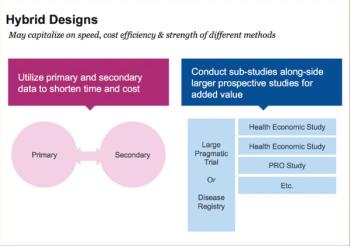

Design analysis can dictate blister composition


Looking at the essential value of well-managed supply chains

Office of Prescription Drug Promotion finds little to complain about

HDMA seeks to encourage more outreach to business partners and government

Despite a flurry of specialty pharmaceuticals in innovative formats, traditional powder-based dosages will always be a mainstay

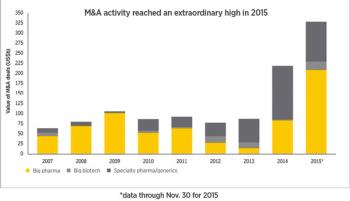
Deal value soared above $300 billion in 2015, but will the dealmaking sustain Big Pharma companies?

Industry is doing better at filing studies, but the agency is falling behind reviews, says GAO

Construction index shows a steady level of North American activity

'Innovation' is the recurring theme

HDMA's 2015-16 Factbook updates member financial, operational performance

Combination expands breadth of commercialization and compliance services

List includes 21 new orphan drugs, and of the 45, 15 are cold chain products

Company partners with Teikoku for the latter's first marketed oncology product, and earns a milestone payment from Teva