
Two Canadian Accucaps facilities join global network of 11 Catalent RP Scherer Softgel locations

Two Canadian Accucaps facilities join global network of 11 Catalent RP Scherer Softgel locations

Working out these details points to how full compliance will look in 2023

HDA survey raises question: what then?

New technologies offer improved shelf life for moisture- and oxygen-sensitive oral solids

Sharpens aim at competitor SAP, while enhancing technical specs
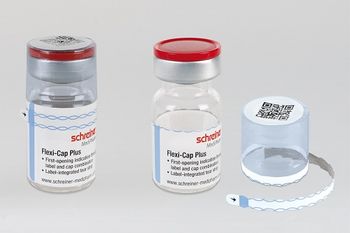
A newly designed label for vials and other containers prevents illicit reuse of pharma containers

Xyntek and Antares Vision build a serialization solution

The growing market for self-administered injectables calls for better patient-training resources

Applying specialty-pharma techniques to a potentially mass-market drug

Vendors and users come together to standardize communication protocols

Company invests $45 million for serialization- and cold-chain-ready capacity

Consumer Products Safety Commission steps into distribution of a generic form of Ambien

As the pharma industry moves ahead with traceability initiatives, vendors are building their capabilities

Packaging designers continue the uphill battle for converting the US pharma industry to unit-dose, compliance-improving packaging
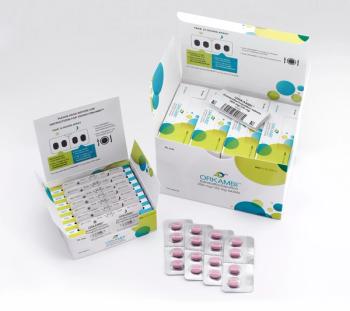
Healthcare Compliance Packaging Council (HCPC) award again goes to PCI Pharma Services

Novo Nordisk and Vetter Pharma announce groundbreakings

An all-compounding "manufacturer" has big plans for growth

Future tech, serialization and South America

Company is now a certified supplier for Walgreens

Pharmaceutical Commerce's annual Cold Chain Sourcebook projects 52% growth between 2014 and 2020

Flexi-Cap Protect prevents breakage at both the tops and bottoms of vials

Quad-C investment will enable future acquisitions

New healthcare proposals re-open talk of drug importation

Government at all levels are trying to mobilize to address the crisis

New survey finds more digitally-savvy healthcare providers, challenging reps' presentation capabilities